 Chromium exhibits +2, +3 and +6 oxidation states. Members of a group typically have similar properties and electron configurations in their outer shell. In all of these scenarios, the new redistribution of the electron is energetically favorable compared to the predicted configuration. The electron configuration of chromium ion(Cr3+) is 1s2 2s2 2p6 3s2 3p6 3d3. Element Chromium (Cr), Group 6, Atomic Number 24, d-block, Mass 51.996. 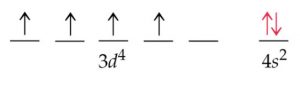 
You will see a similar situation where an #s# electron moves to a #d# sublevel with Molybdenum: So, in this case, an electron moves to #3d# and is unpaired, therefore maximizing the spin state. Since two opposite spins result in a total spin of #0#, maximizing this tends to require as many electrons in of same spin in different orbitals as possible. Hund's Rule: It is energetically favorable to maximize the spin state in a sublevel.The #3d# orbital is slightly lower in energy, and minimizing repulsions in the #4s# orbital by moving one of the #4s# electrons to a close-lying #3d# orbital minimizes the ground-state energy of chromium.It operates on a system of shells and subshells. The electron configuration is a form of notation based on these quantum numbers. This happens in Chromium, as one #4s# electron moves to the #3d# sublevel. Electron Configuration: The electrons of an atom are positioned in its system of atomic orbitals with any single electron position described by a set of four quantum numbers. This actually allows electrons to move between the sublevels if it is energetically favorable. Even though #4s# belongs to a higher main energy level than #3d#, the energy levels are similar. So why is this? It has to do with main the arrangement of sublevels in the electron cloud. However, the actual configuration of Chromium is: Thus, the two electrons in the carbon 2 p orbitals have identical n, l, and m s quantum numbers and differ in their m l quantum number (in accord with the Pauli exclusion principle).Looking at the periodic table, you would expect Chromium to have this configuration: The orbitals are filled as described by Hund’s rule: the lowest-energy configuration for an atom with electrons within a set of degenerate orbitals is that having the maximum number of unpaired electrons. We now have a choice of filling one of the 2 p orbitals and pairing the electrons or of leaving the electrons unpaired in two different, but degenerate, p orbitals. The remaining two electrons occupy the 2 p subshell. Electronic configuration in concert of shell is 2,8,13,1. Four of them fill the 1 s and 2 s orbitals. Electronic configuration of chromium atom is : 1S 2 2S 2 2P 6 3S 2 3P 6 3d 5 4S 1. When drawing orbital diagrams, we include empty boxes to depict any empty orbitals in the same subshell that we are filling.Ĭarbon (atomic number 6) has six electrons. There are three degenerate 2 p orbitals ( m l = −1, 0, +1) and the electron can occupy any one of these p orbitals. Because any s subshell can contain only two electrons, the fifth electron must occupy the next energy level, which will be a 2 p orbital. The n = 1 shell is filled with two electrons and three electrons will occupy the n = 2 shell. The fourth electron fills the remaining space in the 2 s orbital.Īn atom of boron (atomic number 5) contains five electrons. 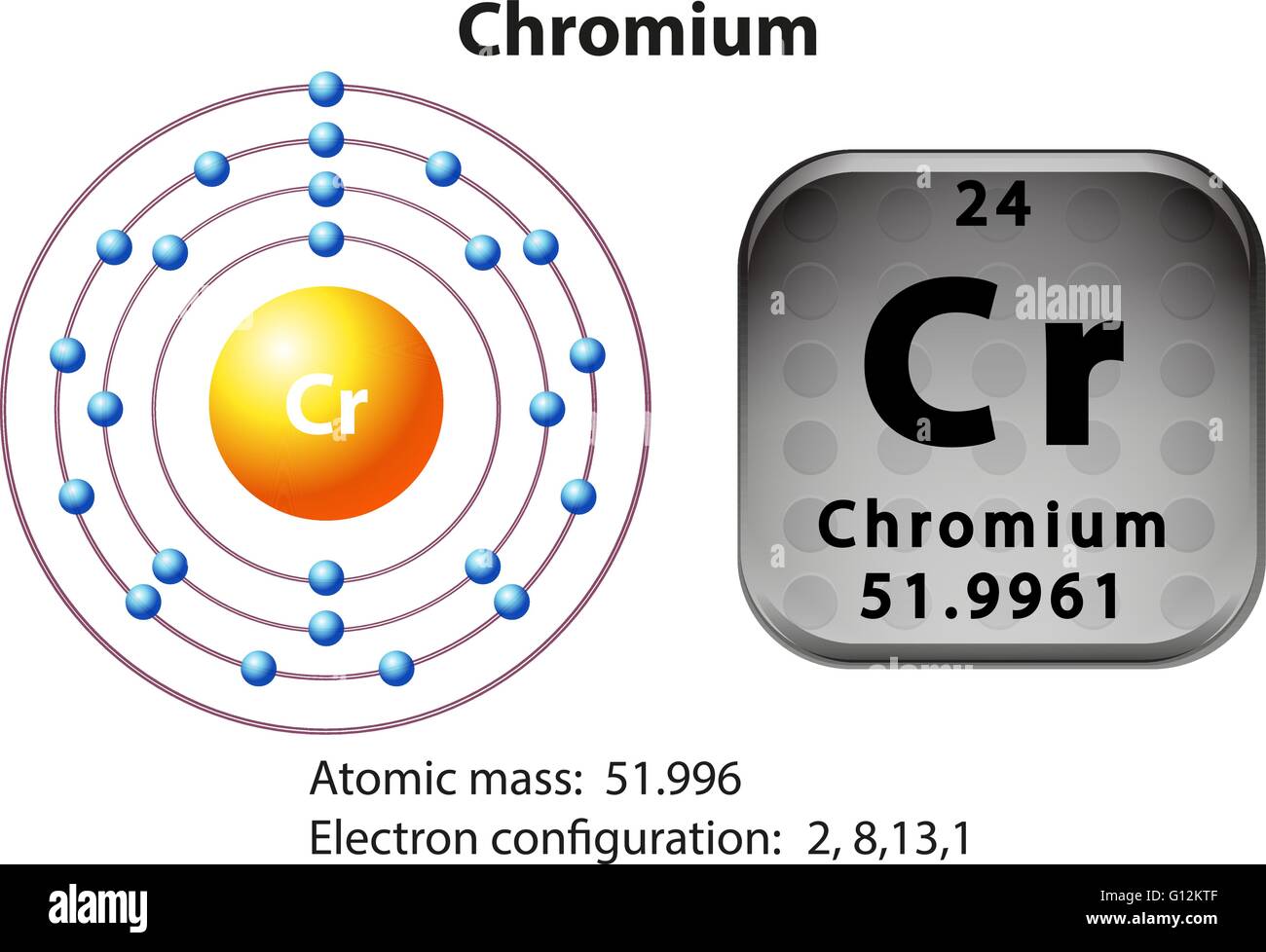
Thus, the electron configuration and orbital diagram of lithium are:Īn atom of the alkaline earth metal beryllium, with an atomic number of 4, contains four protons in the nucleus and four electrons surrounding the nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
